Aortic and coeliac axis compression by the median arcuate ligament
| Available Online: | April, 2023 |
| Page: | 146-148 |
Author for correspondence:
Nada Elzefzaf
Vascular Offices, J Block, The Royal Oldham Hospital, Rochdale Road, OL1 2JH, Oldham, United Kingdom
E-mail: n.elzefzaf@gmail.com
doi: 10.59037/hjves.v4i4.11
ISSN 2732-7175 / 2022 Hellenic Society of Vascular and Endovascular Surgery Published by Rotonda Publications
All rights reserved. https://www.heljves.com
Nada Y. Elzefzaf, Yasmin Oskui, Kathryn A. Lee, Ranjeet Narlawar, Hassan Badri, George A. Antoniou
Department of Vascular Surgery, Manchester University NHS Foundation Trust, Manchester, United Kingdom
Abstract
Full Text
References
Abstract
Median arcuate ligament syndrome is a clinical condition in which the cause of gastrointestinal symptoms is thought to be compression of the coeliac trunk by the median arcuate ligament. We report a case of a 25-year-old woman with compression of the supra-coeliac aorta and the coeliac trunk by the median arcuate ligament. The patient was successfully treated with open surgical decompression. Median arcuate ligament syndrome is poorly understood, and surgical management is highly variable in its outcome.
Keywords: median arcuate syndrome, Dunbar syndrome, coeliac artery compression, unintentional weight loss.
Full Text
INTRODUCTION
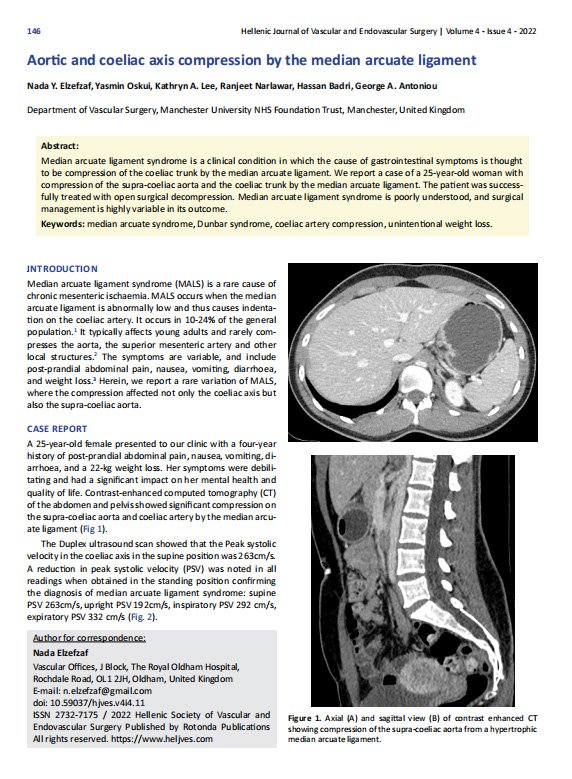
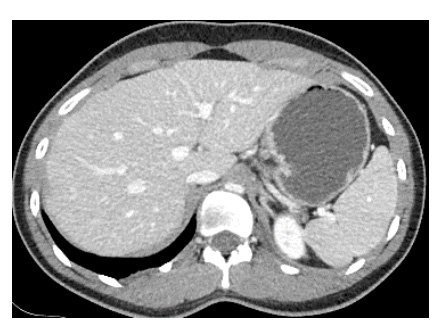
Median arcuate ligament syndrome (MALS) is a rare cause of chronic mesenteric ischaemia. MALS occurs when the median arcuate ligament is abnormally low and thus causes indentation on the coeliac artery. It occurs in 10-24% of the general population.1 It typically affects young adults and rarely compresses the aorta, the superior mesenteric artery and other local structures.2 The symptoms are variable, and include post-prandial abdominal pain, nausea, vomiting, diarrhoea, and weight loss.3 Herein, we report a rare variation of MALS, where the compression affected not only the coeliac axis but also the supra-coeliac aorta.
CASE REPORT
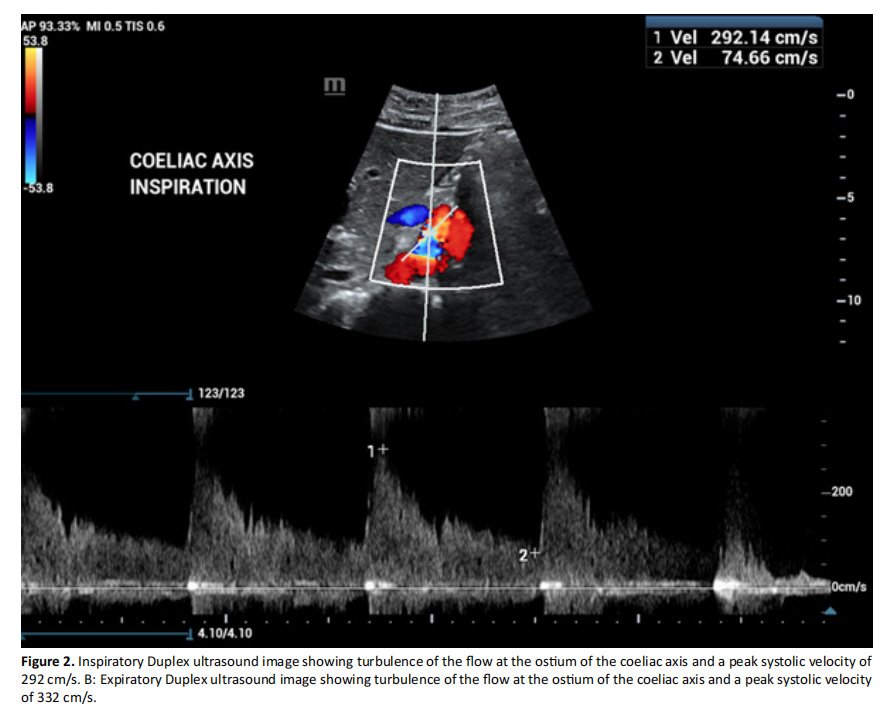
A 25-year-old female presented to our clinic with a four-year history of post-prandial abdominal pain, nausea, vomiting, diarrhoea, and a 22-kg weight loss. Her symptoms were debilitating and had a significant impact on her mental health and quality of life. Contrast-enhanced computed tomography (CT) of the abdomen and pelvis showed significant compression on the supra-coeliac aorta and coeliac artery by the median arcuate ligament (Fig 1). The Duplex ultrasound scan showed that the Peak systolic velocity in the coeliac axis in the supine position was 263cm/s. A reduction in peak systolic velocity (PSV) was noted in all readings when obtained in the standing position confirming the diagnosis of median arcuate ligament syndrome: supine PSV 263cm/s, upright PSV 192cm/s, inspiratory PSV 292 cm/s, expiratory PSV 332 cm/s (Fig. 2).
An open surgical division of the median arcuate ligament and surrounding fibrous bands was performed through a supra-umbilical midline abdominal incision. The superior part of abdominal aorta was dissected and freed from the ligament for around 7cm above the coeliac axis, which was skeletonised up to its bifurcation and released from the ligament and all surrounding fibrotic bands. The patient had an uneventful post operative recovery and was discharged home on day 7. The CT angiogram on day 2 postoperatively revealed reso lution of the compression of the coeliac axis and improvement of the flattened appearance of the supra-coeliac aorta. At her five-week follow-up appointment, she reported reasonable improvement of her symptoms, and had gained 5 kgs since the surgery.
DISCUSSION
MALS was first reported in the mid-1960s 4. The presence of compression was noted in around 34% of autopsies conducted amongst the general population with no reported symptoms, which queries the existence of the condition 5. It occurs more frequently in women (at a ratio of 4:1) 4, 6. The characteristic anatomy in MALS is compression of the coeliac artery by the median arcuate ligament, which fluctuates during inspiration and expiration. The syndrome can also be caused by external compression of other arteries, such as the superior mesenteric artery and the aorta 7. Reilley et described four cases of compression of both the superior mesenteric artery and the coeliac axis 8. Coulier reported a case of a male patient who experienced chronic epigastric pain with aortic compression caused by the median arcuate ligament and no compression of the coeliac trunk or the superior mesenteric artery 9. In our case, there was compression of both the aorta and the coeliac artery. Individuals afflicted with this condition present with gastrointestinal symptoms such as nausea, vomiting, postprandial/exercise induced abdominal pain, and weight loss. The symptoms are indicative of chronic mesenteric ischaemia, and the clinical presentation is variable in its severity 6. In addition to a detailed clinical history, the diagnosis of the condition is established through imaging investigations. There is no gold standard investigation or widely accepted diagnostic criteria for MALS, but the most common imaging investigations include CT angiography and dynamic Duplex ultrasonography. A typical finding on CT is the “hooked” appearance of the coeliac artery. Duplex ultrasonography usually shows elevated velocities in the coeliac axis. Invasive dynamic angiography in inspiration and expiration has been described as a diagnostic tool 10. Treatment of the condition is achieved through surgical decompression of the artery through open, laparoscopic, or robotic release of the median arcuate ligament. The traditional treatment is open surgical decompression, although in recent years, laparoscopic approaches are being increasingly used. Symptomatic relief post-surgery is variable and difficult to predict. Reilly et al. reported the largest series of the longest follow up of open surgical MALS in a total of 51 patients 8. At 10-year follow-up, 53% of patients who had decompression alone had resolution of symptoms compared to 76% of patients who had decompression and revascularization.
CONCLUSION
MALS is a rare clinical condition, and its diagnosis is based on exclusion of other more common pathologies. We believe that the threshold for surgical intervention should be low in such cases as symptom relief is not always achieved, and collaborative experiences from multiple centres may help understand potential links between multi-level disease and severity of symptoms and response to treatment.
References
1 Lainez RA, Richardson WS. Median arcuate ligament syndrome: a case report. Ochsner J 2013;13:561-4.
2 Sunkara T, Caughey ME, Zhen KC, Chiong B, Gaduputi V. Dunbar Syndrome-A Rare Cause of Foregut Ischemia. J Clin Diagn Res 2017;11:OD13-OD14. doi: 10.7860/ JCDR/2017/28142.10267.
3 Sapra A, Franke J, Rahman R, Albers CE, Bhandari P. Fitting the Pieces of the Puzzle Together: A Case of Median Arcuate Ligament Syndrome. Cureus 2021;13:e18384. doi:10.7759/cureus.18384.
4 de Lara FV, Higgins C, Hernandez-Vila EA. Median arcuate ligament syndrome confirmed with the use of intravascular ultrasound. Tex Heart Inst J 2014;41:57-60. doi:10.14503/THIJ-12-2495.
5 Katz-Summercorn A, Bridger J. A cadaveric study of the anatomical variation of the origins of the celiac trunk and the superior mesenteric artery: a role in median arcuate ligament syndrome? Clin Anat 2013;26:971-4. doi:10.1002/ca.22243.
6 Manogna D, Gupta A, Seetharaman M. A Ligamentous Agony: Median Arcuate Ligament Syndrome as an Under-Recognized Cause of Abdominal Pain. Cureus 2020;12:e8865. doi: 10.7759/cureus.8865.
7 Stein JJ, Costanza MJ, Rivero M, Gahtan V, Amankwah KS. External compression of the superior mesenteric artery by the median arcuate ligament. Vasc Endovascular Surg 2011;45:565-7. doi: 10.1177/1538574411412827.
8 Reilly LM, Ammar AD, Stoney RJ, Ehrenfeld WK. Late results following operative repair for celiac artery compression syndrome. J Vasc Surg 1985;2:79-91.
9 Coulier B. Aortic compression by median arcuate ligament during deep inspiration: CT findings. Diagn Interv Imaging 2020;101:425-426. doi: 10.1016/j.diii.2019.09.003.
10 Goodall R, Langridge B, Onida S, Ellis M, Lane T, Davies Median arcuate ligament syndrome. J Vasc Surg 2020;71:2170-2176. doi: 10.1016/j.jvs.2019.11.012.

